PhysioNet
(Physiology & Physiopathology of Brain Networks Team)
(Physiology & Physiopathology of Brain Networks Team)
PhysioNet is an interdisciplinary team of researchers pooling expertise from diverse backgrounds (engineering, biology, physics, medicine, mathematics) and aiming to understand the physiology of biological neuronal networks.
There are currently three research groups in PhysioNet whose activities overlap and that tackle the understanding of neuronal network physiology at multiple levels, from cellular physiology with patch clamp to neural population recording with extracellular electrophysiology, to whole-brain imaging and modelling.
EMAIL: pascale.quilichini@univ-amu.fr
PHONE: +33 4 91 32 42 31
—————————————————————————-
Shunzuke KAJIWARA | PostDoc
Myriam AZZARELLI | PhD Student
Richard BOYCE | PostDoc
Monique ESCLAPEZ | DR Inserm
Ouafae ARAB | AMU IR
Maëva FERRARIS | PhD Student/PostDoc
Thomas DOUBLET | PostDoc
Armelle LOKOSSOU | PostDoc
Matthias DIPPER-WAWRA | PostDoc
Wesley CLAWSON | PhD Student
Yoshi NAKATANI | PhD Student
Ana FERNANDEZ VICENTE | PostDoc
Galyna MALIEIEVA | PhD Student/PostDoc
Priya GHUMATKAR | PostDoc
Loïg KERGOAT | PostDoc
Francesca MELLOZI | PhD Student
…
Nathan MINOUNI | Centrale-Supélec Paris
Correntin LASNE | Centrale-Supélec Paris
Yann ROCHE | Centrale-Supélec Paris
Raphaël NUNES DA SILVA | Centrale-Supélec Paris
Hugo DEGENEVE | Centrale-Supélec Paris
Valentin GRISEL | Epitech Marseille
Sarah WU | Centrale-Supélec Paris
Paul-Arno LAMARQUE | Centrale-Supélec Paris
Andrea Nuti | Scuola Superiore Sant’ Anna
Noé Hamou | ESPCI Paris
Benjamin WAKED | Centrale-Supélec Paris
Etienne GUEVEL | Centrale-Supélec Paris
Omar BENJELLOUN | ESPCI Paris
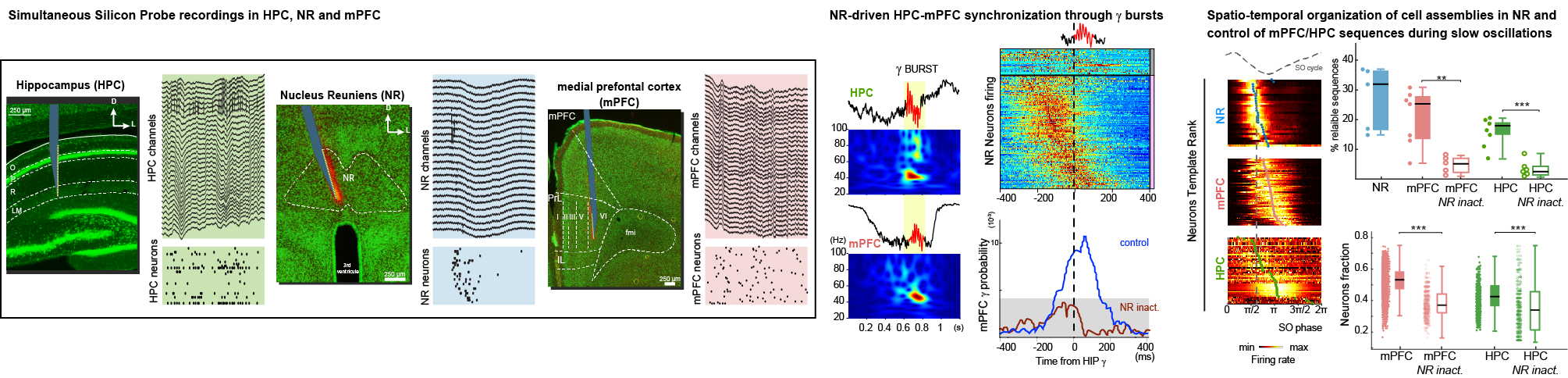
Cognitive processes depend upon the activity of distributed networks in the brain. We use multiple silicon probes recordings in rodents sampling the network activities and the firing of a large number of neurons in the hippocampus, prefrontal cortex, entorhinal cortex and thalamic nucleus reuniens.
Our two main goals are:
(1) Understand the fundamental mechanisms underlying the communication between brain regions controlling memory processes by deciphering how cortico-thalamo-hippocampal networks exchange information to encode and consolidate spatial-related information.
(2) Understand how these physiological rules are modified in different pathological conditions, such as epilepsy.
We use data mining approaches to determine how the dynamics of hundreds of individual neurons and local network oscillations collected in animals performing memory-related tasks can support both memory function and dysfunction.

Whatever it is, the way you tell your story online can make all the difference.
We are interested in the dynamics allowing the exchange of information between brain areas, namely how the information encoded in one neural structure is able to modulate the encoding in another area (and the other way around), how these interactions orchestrate perception, learning, and behavior, and how these mechanisms are affected by epilepsy. To do so, our primary experimental approach is to perform high-density recordings of individual neurons simultaneously in multiple brain sites in freely behaving rats and then use data mining and analysis to study neural population dynamics.
Using mathematical and modeling approaches in close collaboration with the TNG team of V. Jirsa, we are studying the basic mechanisms of seizure genesis and propagation across species. We also investigate the mechanisms of vulnerability to epilepsy induced by stress as well as the co-morbidities such as depression, cognitive deficits (memory). We use a multi-disciplinary approach in healthy animals (rats and mice), experimental models of epilepsy (pilocarpine and kainite models) and Alzheimer (APPNL-G-F transgenic mice) in which we couple multisite recordings (in close collaboration with P. Quilichini group) and behavior.

Whatever it is, the way you tell your story online can make all the difference.
Figures extracted from:
Ferraris et al (2018) J Neurosci 38(12):3026-3038. & Angulo-Garcia et al (2020) J Neurosci 40:8343-835.
Clawson et al (2023) J Neurosci 43(38: 6573-6587.
Ghestem et al (2023) J. Neural Eng. 20:046003.
Makdah G, Wiener SI, Pompili MN (2025) Detection of Cell Assemblies in High-Density Extracellular Electrophysiological Recordings. In: Carrillo-Reid, L. (eds) “Identification, Characterization, and Manipulation of Neuronal Ensembles“. Neuromethods vol 215. Springer
Rabuffo G, Lokossou A, Li Z, Ziaee-Mehr A, Hashemi M, Quilichini PP, Ghestem A, Arab O, Esclapez M, Verma P, Raj A, Gozzi A, Sorrentino P, Chuang K, Perles-Barbacaru T, Viola A, Jirsa V, Bernard C (2025) Mapping global brain reconfigurations following local targeted manipulations. PNAS 122(16): e2405706122
Pompili MN, Todorova R, Boucly C, Leroux E, Wiener SI, Zugaro M (2025) Adaptive communication between cell assemblies and ‘reader' neurons shapes flexible brain dynamics. PLoS biology 23(12): e3003505
Pompili MN, Eckmier A, Tirole M, Todorova R, Godsil BP, Jay TM (2025) Ventral Hippocampus Modulates Prefrontal Control of Background Contextual Fear After Cued Extinction. European Journal of Neuroscience 62(8): e70287
Mercier O, Quilichini PP, Magalon K, Gil F, Ghestem A, Richard F, Boudier T, Cayre M, Durbec P. (2024) Transient demyelination causes long‐term cognitive impairment, myelin alteration and network synchrony defects. Glia 72 (5), 960-981
Clawson W, Waked B, Madec T, Ghestem A, Quilichini P.P, Battaglia D, Bernard C. (2023). Perturbed Information Processing Complexity in Experimental Epilepsy. J Neurosci 43 (38) 6573-6587. doi: 10.1523/JNEUROSCI.0383-23.2023
Bernard, C, Frauscher, B, Gelinas, J, Timofeev, I. (2023) Sleep, oscillations, and epilepsy. Epilepsia 00: 1–10. doi: 10.1111/epi.17664
Ghestem A, Pompili MN, Dipper-Wawra M, Quilichini PP, Bernard C, Ferraris M (2023) Long-term near-continuous recording with Neuropixels probes in healthy and epileptic rats. Journal of Neural Engeneering 20 046003. doi: 10.1088/1741-2552/ace218.
Doublet T, Ghestem A, Bernard C (2022) Deficit in observational learning in experimental epilepsy. Epilepsia 63 (12), e150-e155. doi: 10.1111/epi.17421
Pompili MN, Todorova R (2022) Discriminating sleep from freezing with cortical spindle oscillations. Frontiers in Neural Circuits 16: 783768
Rabuffo G, Sorrentino P, Bernard C, Jirsa V (2022) Spontaneous neuronal avalanches as a correlate of access consciousness. Frontiers in Psychology 13, 1008407. doi:
Demars F, Todorova R, Makdah G, Godsil BP, Krebs MO, Jay TM, Wiener SI, Pompili MN (2022) Post-trauma behavioral phenotype predicts the degree of vulnerability to fear relapse after extinction in male rats. Current Biology 32(14): 3180-3188
Brancati GE, Rawas C, Ghestem A, Bernard C, Ivanov AI. (2021) Spatio-temporal heterogeneity in hippocampal metabolism in control and epilepsy conditions. Proc Natl Acad Sci U S A. 118(11):e2013972118. doi: 10.1073/pnas.2013972118.
Ferraris M, Cassel JC, Pereira de Vasconcelos A, Stephan A, Quilichini PP. (2021) The nucleus reuniens, a thalamic relay for cortico-hippocampal interaction in recent and remote memory consolidation. Neurosci Biobehav Rev. 125:339-354. doi: 10.1016/j.neubiorev.2021.02.025.
Cassel JC, Ferraris M, Quilichini P, Cholvin T, Boch L, Stephan A, Pereira de Vasconcelos A. (2021) The reuniens and rhomboid nuclei of the thalamus: A crossroads for cognition-relevant information processing? Neurosci Biobehav Rev. 126:338-360. doi: 10.1016/j.neubiorev.2021.03.023.
Gomez-Castro F, Zappettini S, Pressey JC, Silva CG, Russeau M, Gervasi N, Figueiredo M, Montmasson C, Renner M, Canas P, Gonçalves FQ, Alçada-Morais S, Szabó E, Rodrigues RJ, Agostinho P, Tomé AR, Caillol G, Thoumine O, Nicol X, Leterrier C, Lujan R, Tyagarajan SK, Cunha RA, Esclapez M, Bernard C, Lévi S (201) Convergence of adenosine and GABA signaling for synapse stabilization during development. Science 374, eabk2055. doi: 10.1126/science.abk2055
Pedreschi N, Bernard C, Clawson W, Quilichini P, Barrat A, Battaglia D. (2020) Dynamic core-periphery structure of information sharing networks in entorhinal cortex and hippocampus. Network Neurosci. 4(3):946-975. doi: 10.1162/netn_a_00142.
Debski KJ, Ceglia N, Ghestem A, Ivanov AI, Brancati GE, Bröer S, Bot AM, Müller JA, Schoch S, Becker A, Löscher W, Guye M, Sassone-Corsi P, Lukasiuk K, Baldi P, Bernard C (2020). The circadian dynamics of the hippocampal transcriptome and proteome is altered in experimental temporal lobe epilepsy. Science advances, 6(41), eaat5979. doi: 10.1126/sciadv.aat5979
Angulo-Garcia D, Ferraris M, Ghestem A, Nallet-Khosrofian L, Bernard C, Quilichini PP (2020) Cell Assemblies in the Cortico-Hippocampal-Reuniens Network during Slow Oscillations. J Neurosci 40:8343-835. doi: 10.1523/JNEUROSCI.0571-20.2020
Vicente AF, Slézia A, Ghestem A, Bernard C, Quilichini PP. (2020) In Vivo Characterization of Neurophysiological Diversity in the Lateral Supramammillary Nucleus during Hippocampal Sharp-wave Ripples of Adult Rats. Neuroscience. 2020; 435:95-111. doi: 10.1016/j.neuroscience.2020.03.034.
Saggio ML, Crisp D, Scott JM, Karoly P, Kuhlmann L, Nakatani M, Murai T, Dümpelmann M, Schulze-Bonhage A, Ikeda A, Cook M, Gliske SV, Lin J, Bernard C, Jirsa V, Stacey WC (2020) A taxonomy of seizure dynamotypes. Elife 9:e55632. doi: 10.7554/eLife.55632.
Billwiller F, Castillo L, Elseedy H, Ivanov AI, Scapula J, Ghestem A, Carponcy J, Libourel PA, Bras H, Abdelmeguid NE, Krook-Magnuson E, Soltesz I, Bernard C, Luppi PH, Esclapez M (2020) GABA-glutamate supramammillary neurons control theta and gamma oscillations in the dentate gyrus during paradoxical (REM) sleep. Brain Struct Funct. 225(9):2643-2668. doi: 10.1007/s00429-020-02146-y.
Melozzi F, Bergmann E, Harris JA, Kahn I, Jirsa V, Bernard C (2019) Individual structural features constrain the mouse functional connectome. Proc Natl Acad Sci U S A. 116(52):26961-26969. doi: 10.1073/pnas.1906694116.
Clawson W, Vicente AF, Ferraris M, Bernard C, Battaglia D, Quilichini PP. (2019) Computing hubs in the hippocampus and cortex. Sci Adv 5(6): eaax4843, doi: 10.1126/sciadv.aax4843.
Manouze H, Ghestem A, Poillerat V, Bennis M, Ba-M'hamed S, Benoliel JJ, Becker C, Bernard C (2019) Effects of Single Cage Housing on Stress, Cognitive, and Seizure Parameters in the Rat and Mouse Pilocarpine Models of Epilepsy. eNeuro 6. doi: 10.1523/ENEURO.0179-18.2019
Baud MO, Ghestem A, Benoliel JJ, Becker C, Bernard C (2019) Endogenous multidien rhythm of epilepsy in rats. Exp Neurol 315:82-87. doi: 10.1016/j.expneurol.2019.02.006
Ferraris M, Ghestem A, Vicente AF, Nallet-Khosrofian L, Bernard C, Quilichini PP. (2018) The Nucleus Reuniens Controls Long-Range Hippocampo-Prefrontal Gamma Synchronization during Slow Oscillations. J Neurosci. 38(12):3026-3038. doi: 10.1523/JNEUROSCI.3058-17.2018.
Frigerio F, Flynn C, Han Y, Lyman K, Lugo JN, Ravizza T, Ghestem A, Pitsch J, Becker A, Anderson AE, Vezzani A, Chetkovich D, Bernard C. (2018) Neuroinflammation Alters Integrative Properties of Rat Hippocampal Pyramidal Cells. Mol Neurobiol. Sep;55(9):7500-7511. doi: 10.1007/s12035-018-0915-1.